Do anti-cholinergic drugs cause dementia?
The experts... are wrong?
Anti-cholinergic drugs make you dumber, sleepier, and clumsier by blocking acetylcholine, a neurotransmitter involved in motor function and memory. This aggrevates the symptoms of dementia, but the effect subsides once the drug leaves your body.
Beyond that, there is a scientific consensus that they cause dementia in the long term.
It’s incorrect.
This is one of the biggest academic blind spots I’ve seen in my life. Anti-cholinergics are not a well-known drug class, but they are taken by about 25% of the general population; more common than anti-depressants.
These drugs are thought to cause dementia for multiple reasons. One of them is that some anti-cholinergics block1 the M1 receptor, which can then cause the brain to produce less α-secretase and more α/β peptide; α/β peptide accumulates into amyloid plaques, which cause cognitive decline. Essentially:
Drug → M1 receptor block→ more α/β peptide →amyloid plaques → cognitive decline (specifically Alzheimer’s)
The amyloid hypothesis of Alzheimer’s has attracted some doubt. One of the researchers who published papers that influenced the theory was found to have committed tons of fraud; some of the papers were retracted. Prior to this finding, there were people who were skeptical of the theory, noting that many of the drugs based on it failed FDA trials, while the others that were approved were not related to it.
The presence of amyloid plaque is still a pretty strong predictor of the disease. There is almost no overlap in the amount of plaque in Alzheimer’s patients and similarly aged controls who have no dementia. At least, in some parts of the brain:
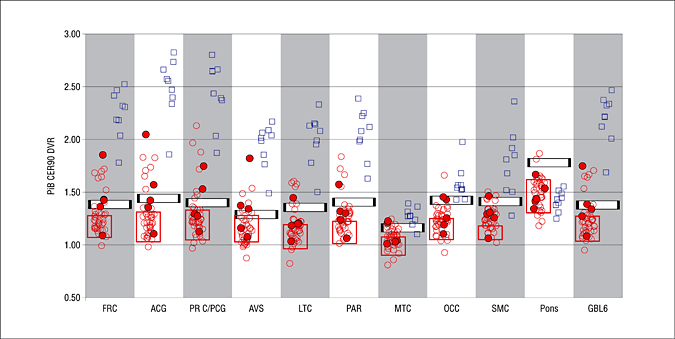
It seems that the plaques themselves don’t cause cognitive decline, it’s the tau tangles. One study reports baseline amyloid plaque levels had no statistically significant association with declines in future test scores, but tau tangles did:
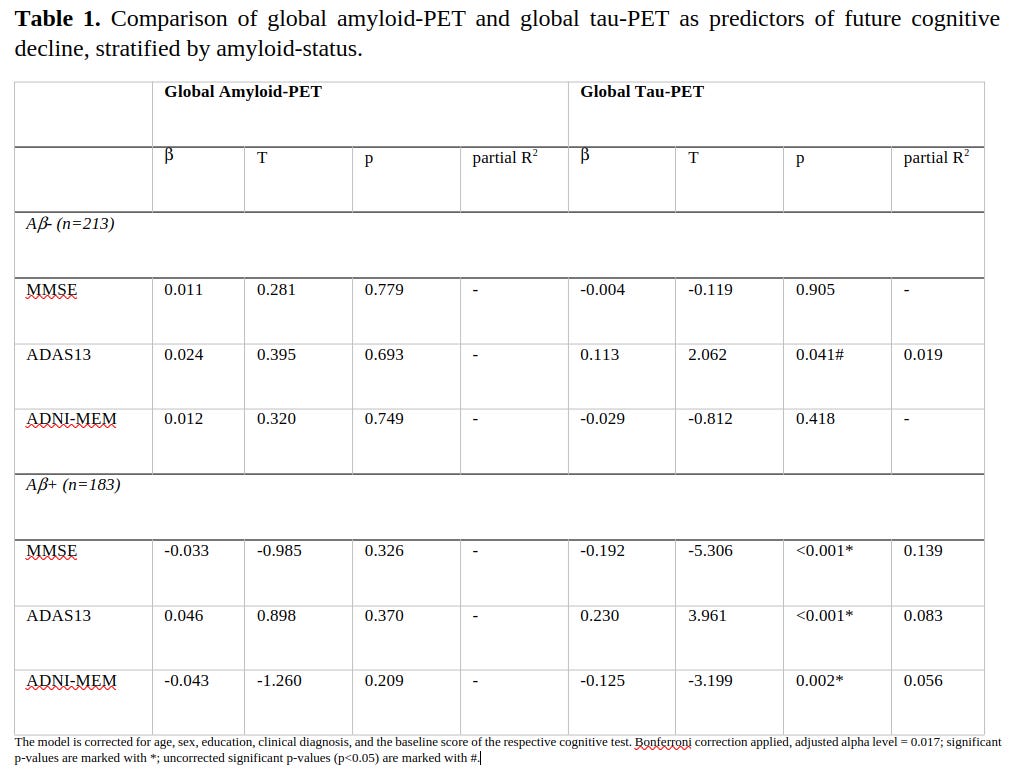
The statistical reasoning here is faulty. Within people who had elevated amyloid plaque levels (A/β+), the association between plaque and cognitive decline was not statistically significant, but it was in the expected direction. They claimed to correct the association between tau tangle baseline levels and cognitive decline for plaque levels in their regression model, but it doesn’t look to me like they did.
It seems that, at the early stage of Alzheimer’s disease, α/β plaques are more present than tau tangles, and both predict the future levels of tau tangles. Once the disease gets going, plaque becomes less relevant and tau starts dominating2:
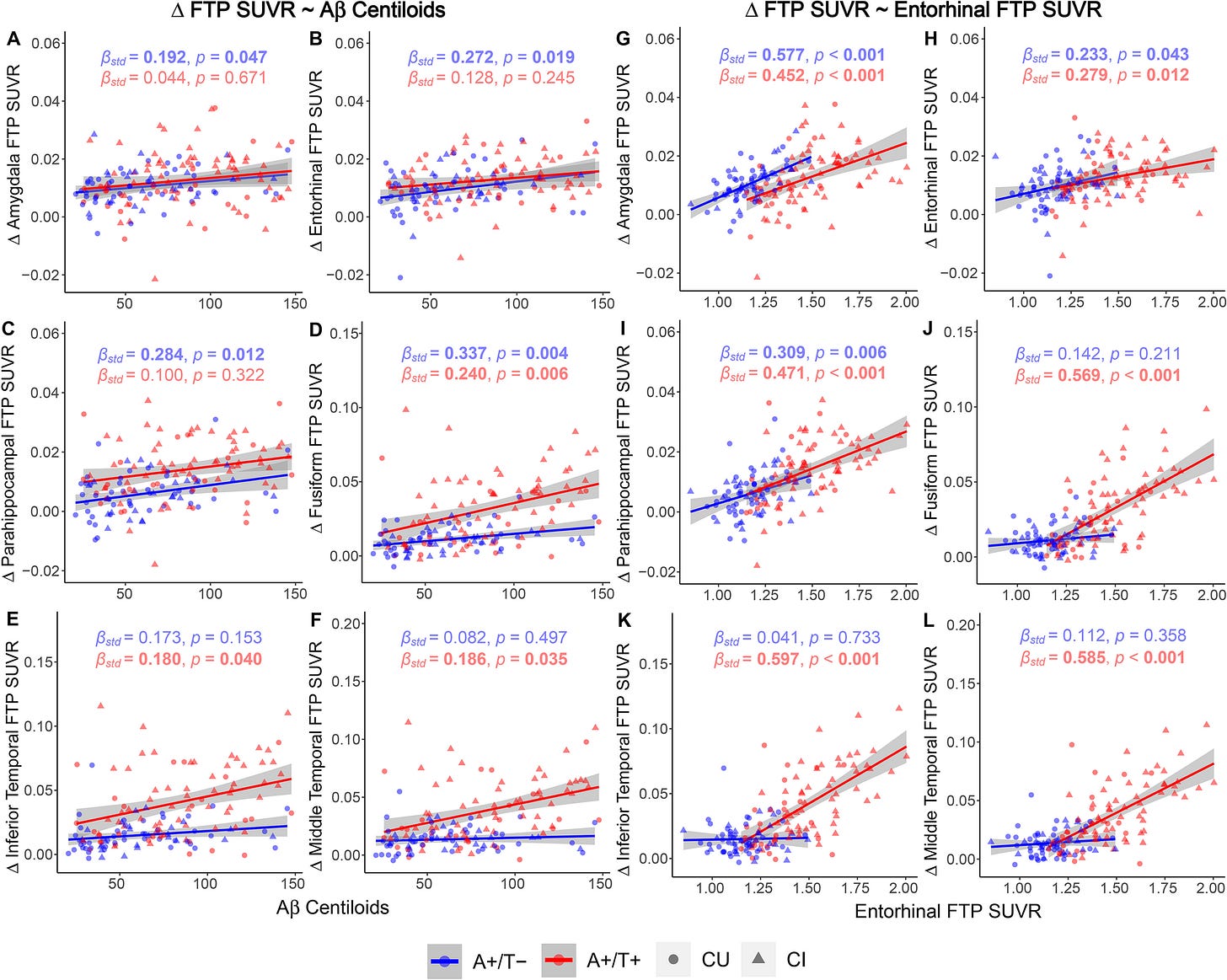
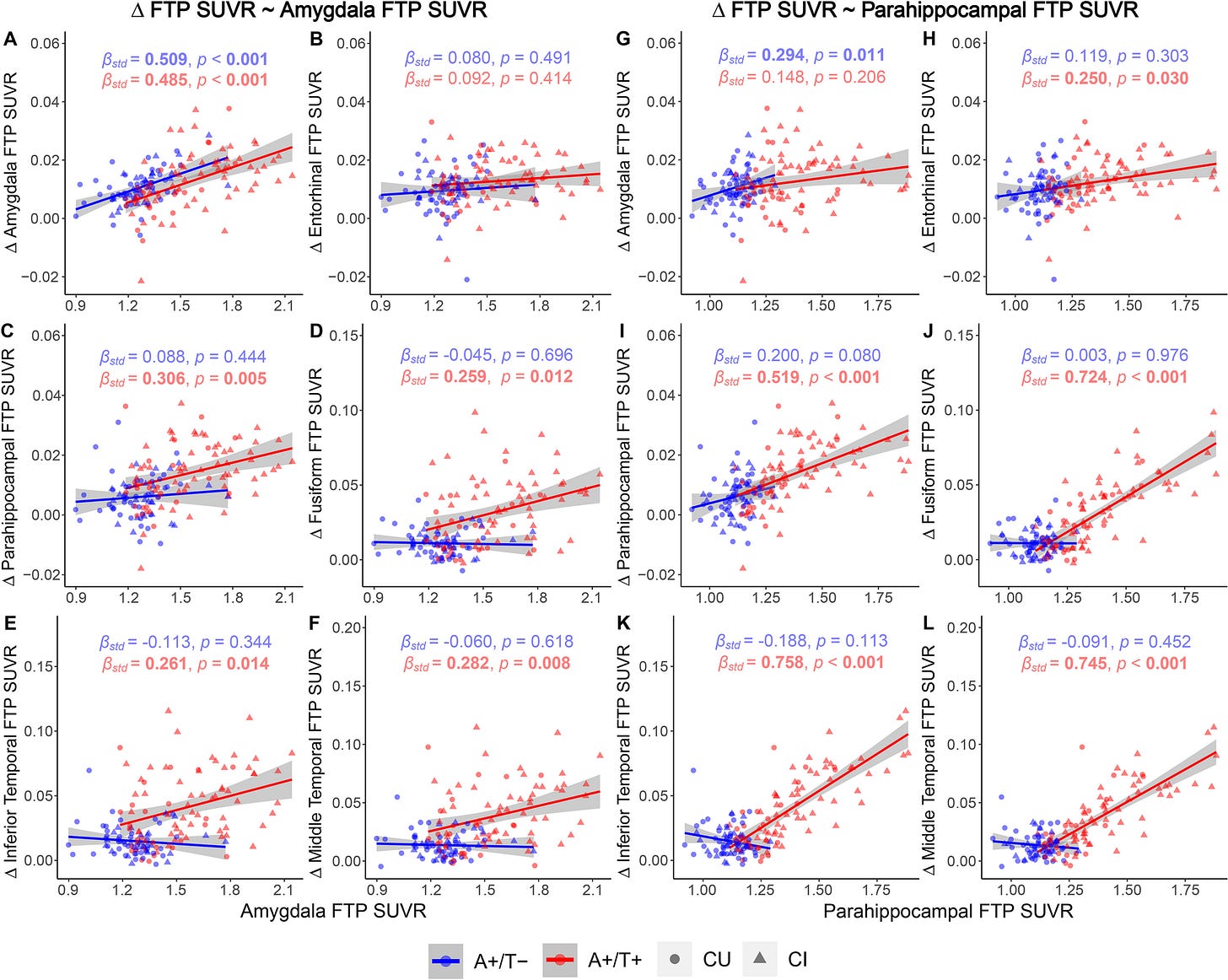
I suppose this explains why drugs that were developed based on the theory were so unsuccessful. By the time somebody is diagnosed with dementia, they already have plaques and tau tangles in their brain. Preventing the creation of plaque doesn’t deal with the tau tangles that are directly driving the cognitive decline.
Overall, the plaque/tau theory of Alzheimer’s holds up reasonably well to scrutiny.
There are also a few other mechanisms by which anti-cholinergic drugs could affect dementia. People adapt to the effects of anti-cholinergic drugs by upregulating the receptors that are blocked and/or increasing their sensitivity; if we believe the plaque/tau hypothesis, then the tolerance effect should theoretically counterbalance some of the effect of M1 blockage.
Use of anti-cholinergics could also lead to selective synaptic pruning, where cholinergic neurons are selectively disconnected by the brain due to lower activity. Not implausible, but hard to test.
The hypothesis that these drugs cause dementia is sensible in the way the diet-heart theory is. It’s rational, but some of the theoretical mechanisms by which they do involve lots of mechanistic causal chains, which imply an uncertain and attenuated effect size.
What I mean by this, is that if A has a 0.5 SD effect on B, and B has a 0.5 SD effect on C, the effect of A on C is 0.25; not 0.5. In this hypothesis, there are a total of five steps between anti-cholinergic drug intake and cognitive decline: drugs → M1 blockage → α/β peptide upregulation →formation of plaques →tau protein accumulation →cognitive decline. If any one of these effects don’t exist, or if the drugs have effects in the opposite direction that counteract the main chain, the theory flies out the window.
Oh, and those studies people cite to argue that M1 receptor blockage is associated with α/β peptide and amyloid plaque? Most of it is literally in mice. I found one human study that finds M1 agonists reduce AB peptide levels by 20%; it’s a p < .004 with 19 people. I guess that’s not terrible.
When it comes to observational data on this topic, this study stands out as being particularly large and high quality:
Coupland CAC, Hill T, Dening T, Morriss R, Moore M, Hippisley-Cox J. Anticholinergic Drug Exposure and the Risk of Dementia: A Nested Case-Control Study. doi: 10.1001/jamainternmed.2019.0677
Of the entire study population (284 343 case patients and matched controls), 179 365 (63.1%) were women, and the mean (SD) age of the entire population was 82.2 (6.8) years. The adjusted OR for dementia increased from 1.06 (95% CI, 1.03-1.09) in the lowest overall anticholinergic exposure category (total exposure of 1-90 TSDDs) to 1.49 (95% CI, 1.44-1.54) in the highest category (>1095 TSDDs), compared with no anticholinergic drug prescriptions in the 1 to 11 years before the index date. There were significant increases in dementia risk for the anticholinergic antidepressants (adjusted OR [AOR], 1.29; 95% CI, 1.24-1.34), antiparkinson drugs (AOR, 1.52; 95% CI, 1.16-2.00), antipsychotics (AOR, 1.70; 95% CI, 1.53-1.90), bladder antimuscarinic drugs (AOR, 1.65; 95% CI, 1.56-1.75), and antiepileptic drugs (AOR, 1.39; 95% CI, 1.22-1.57) all for more than 1095 TSDDs. Results were similar when exposures were restricted to exposure windows of 3 to 13 years (AOR, 1.46; 95% CI, 1.41-1.52) and 5 to 20 years (AOR, 1.44; 95% CI, 1.32-1.57) before the index date for more than 1095 TSDDs. Associations were stronger in cases diagnosed before the age of 80 years. The population-attributable fraction associated with total anticholinergic drug exposure during the 1 to 11 years before diagnosis was 10.3%.
Their results:
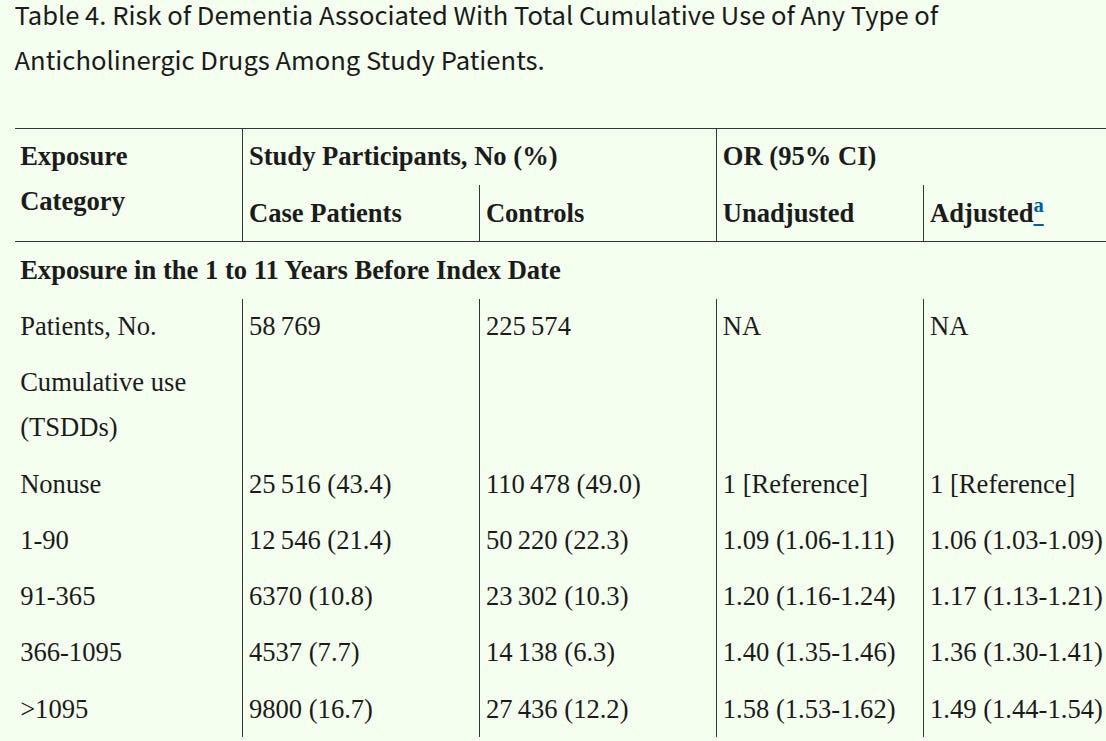
Pretty unambiguous as far as statistics go, the lower 95% CIs are far away from 1.
The elephant in the room: people experiencing cognitive decline need medications to control the symptoms: insomnia, psychosis, urinary incontinence, and such. It just so happens that a lot of the drugs prescribed to treat these symptoms are also anti-cholinergics, so it’s not surprising the users are more likely to experience dementia later.
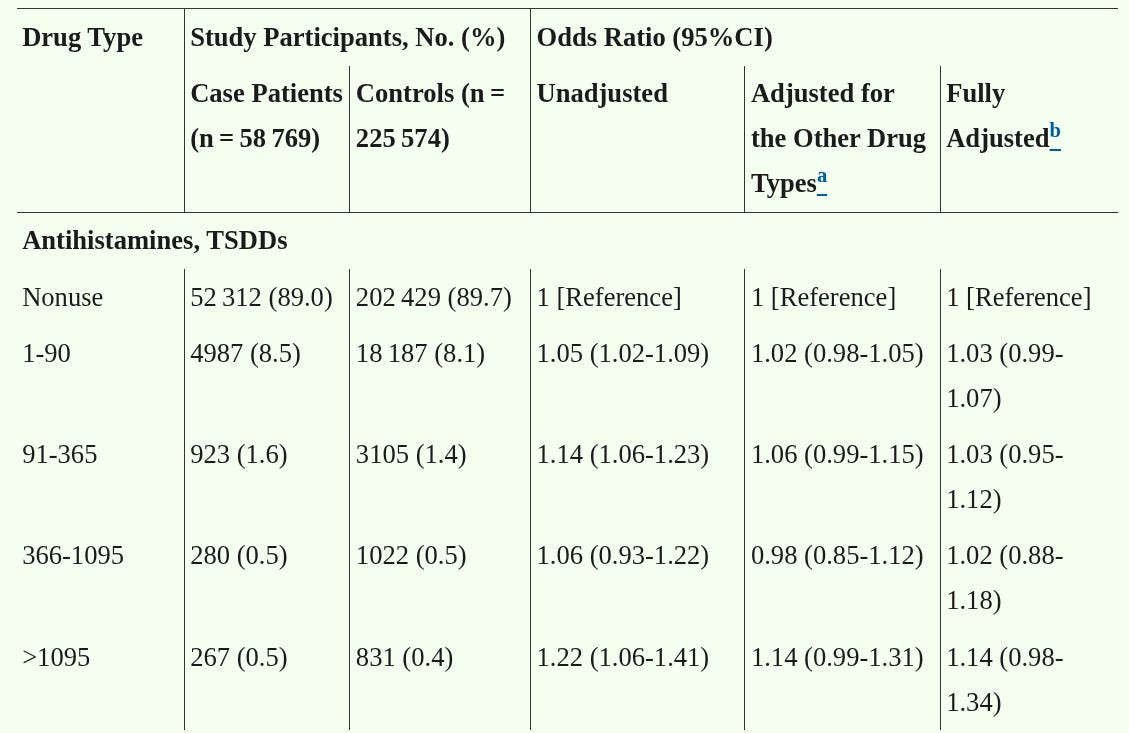
The other issue is that not all of the anti-cholinergics were even associated with future cognitive decline. The anti-histamine variants were hardly correlated with future cognitive decline, as were the antiemetics, muscle relaxants, gastro antispasmodics, antiarrythmics, and bronchodilators — when you adjusted for the effects of taking other drugs.
I’ve seen it argued that this is because the “strong anti-cholinergics” are the ones that cause dementia, but the weaker ones do not. This is not borne out in the data. For example, benadryl is strongly anti-cholinergic — but the other anti-histamine drugs they lumped it with are not associated with cognitive decline.
Perhaps they accidentally lumped benadryl with anti-histamines that are less potent or do not get into the brain’s blood, so the real effect got lost in the mess. Here’s the problem: every single anti-histamine that they used in their study — Azatadine, Brompheniramine, Chlorpheniramine, Clemastine, Cyproheptadine, Hydroxyzine, Trimeprazine — was a first generation variant that does cross into the brain. The fact these drugs are not assoicated with dementia is quite telling.
Researchers try to overcome this with case-controls, examining long term use before diagnoses, or controlling for demographic/SES variables. “Controlling for variables” is overly maligned in statistics, but this is a uniquely egregious case — they aren’t controlling for the thing that matters: existing cognitive decline and genetic/environmental predisposition.
Cognitive decline is also a long term process. It can start as early as people’s 30s:

I checked the drugs they evaluated in that observational study: some of them don’t even cross the blood-brain barrier. Glycopyrrolate (bronchodilator), Ipratropium (bronchodilator), Propantheline (gastro antispasmodic), Trospium (bladder control drug), Flavoxate (bladder control drug), and Butylscopolamine (gastro antispasmodic) are all below-the-neck drugs.
Out of the gate, the anti-cholinergic drug hypothesis looks sketchy.
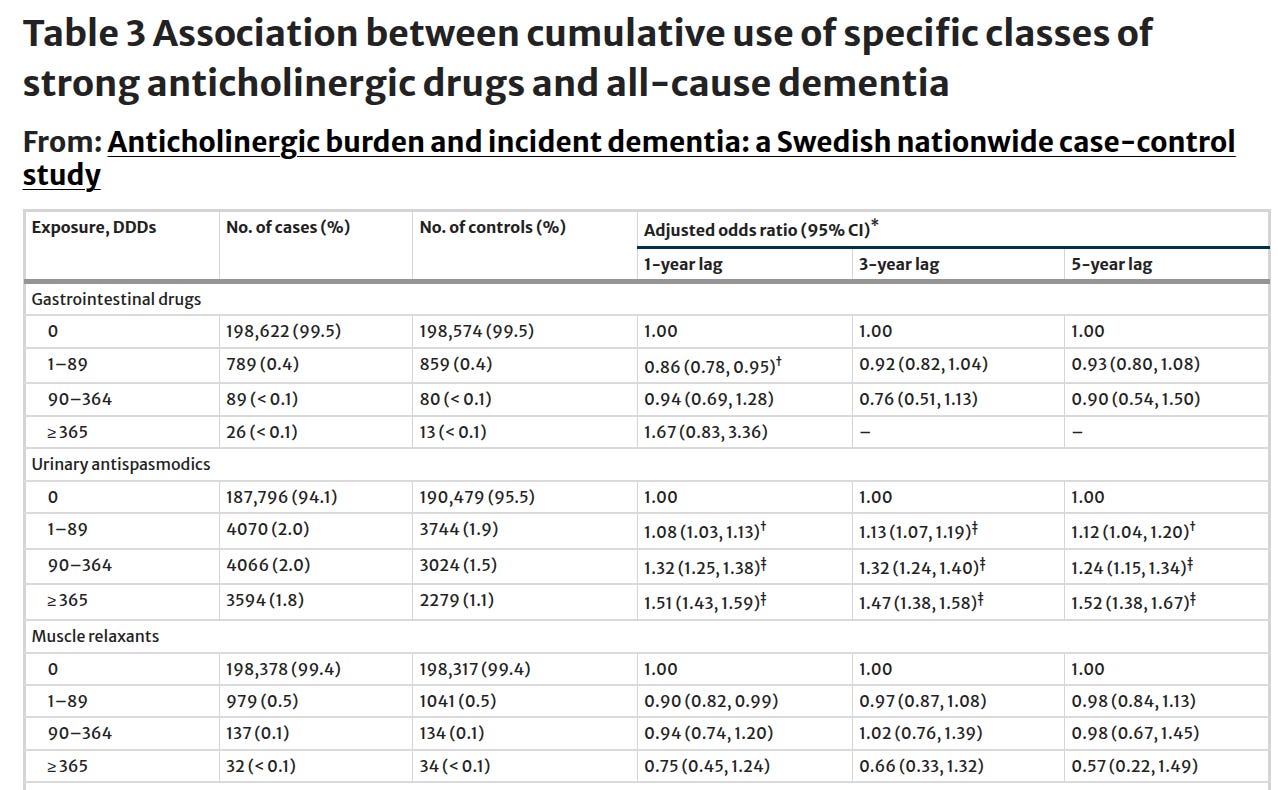
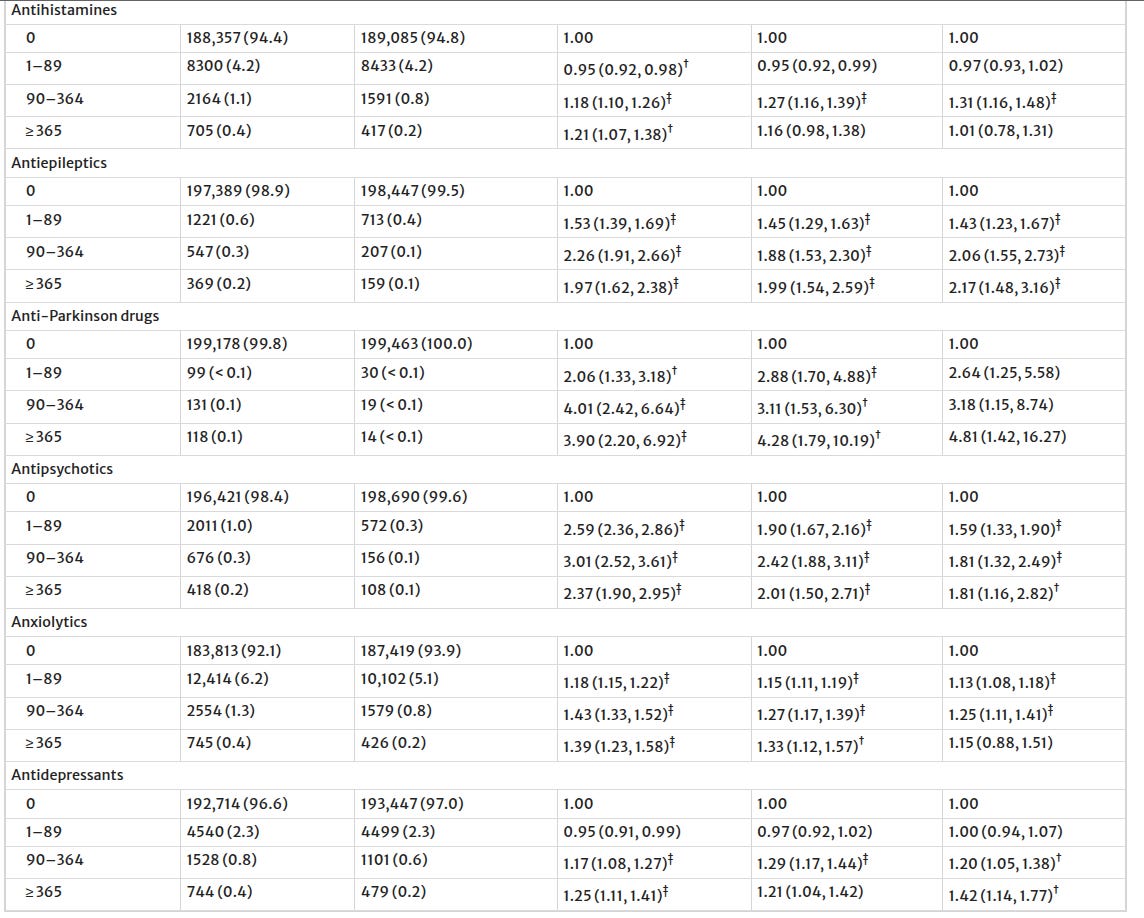
I found a similar Swedish study that was published recently, focused on vascular dementia. I looked at their table3, and it turns out they found the same thing: anti-depressants, anti-spasmodics, anti-epileptics, anti-psychotics, and anti-parkinsons drugs with anti-cholinergic properties were all associated with cognitive decline; everything else was a null.
A nonlinear dose-response relationship with all-cause dementia was found for the cumulative use of strong anticholinergics (e.g., 1–89 DDDs: AOR, 1.10 [95% CI, 1.08–1.12]; ≥1095 DDDs: AOR, 1.66 [95% CI, 1.55–1.79]), notably within the class of urinary antispasmodics, antihistamines, and psychotropic drugs. In contrast, a subtle association lacking a dose-response pattern was observed for weak anticholinergics (e.g., 1–89 DDDs: AOR, 1.11 [95% CI, 1.08–1.13]; ≥1095 DDDs: AOR, 1.01 [95% CI, 0.98–1.03]). The associations with strong anticholinergics were more pronounced in men, younger people, those diagnosed with vascular dementia or Lewy body dementia (versus Alzheimer’s disease), and those with milder-stage dementia.
Consistent with the drug-specific effects found earlier. Out of curiosity, I checked the UKBB study on the same topic: it found associations between dementia and anti-depressants, anti-epileptics, diuretics; no for everything else. Antipsychotics had a positive, but statistically insignificant relationship.
Coincidentally, it seems like all of the drugs you would expect somebody experiencing early-onset cognitive decline — anhedonia (treated with anti-depressants), psychosis (treated with anti-psychotics), loss of dexterity (trated with anti-parkinson’s), and urinary issues (treated with anti-spasmodics) — to be taking, all just happen to be associated with future dementia.
People have tried testing the theory in more creative ways: one of them involved examining the effect of taking hypertensive drugs that had anti-cholinergic side effects on dementia. They found nothing.
Studying the effect of duration of treatment on prognostic outcomes using real-world data is challenging because only people who survive for a long time can receive a treatment for a long time. Specifying a target trial helps overcome such challenge. We aimed to estimate the effect of different durations of treatment with antihypertensive drugs with anticholinergic properties (AC AHT) on the risk of vascular dementia and Alzheimer's disease by emulating a target trial using the UK CPRD GOLD database (2001-2017). Comparing treatment for 3-6 years vs ≤3 years yielded null results for both types of dementia. Comparing a longer duration of treatment, >6 years vs ≤3 years, yielded a 10-year risk ratio of 0.69 (95% CI, 0.54-0.90) for vascular dementia and 0.91 (95% CI, 0.77-1.10) for Alzheimer's disease. For illustration, we performed an analysis that failed to emulate a target trial by assigning exposure categories using postbaseline information, obtaining implausible beneficial estimates. Our findings indicate a modest benefit of longer duration of treatment with AC AHT on vascular dementia and highlight the value of the target trial emulation to avoid selection bias in the evaluation of the effect of different durations of treatment. This article is part of a Special Collection on Pharmacoepidemiology.
If anything, it looks like the drugs helped. Which isn’t surprising, since the cardiovascular system is also implicated in cognitive decline. It led to a stronger reduction in vascular dementia4, which is caused by repeated strokes; the effect on Alzheimer’s was protective but not statistically significant.
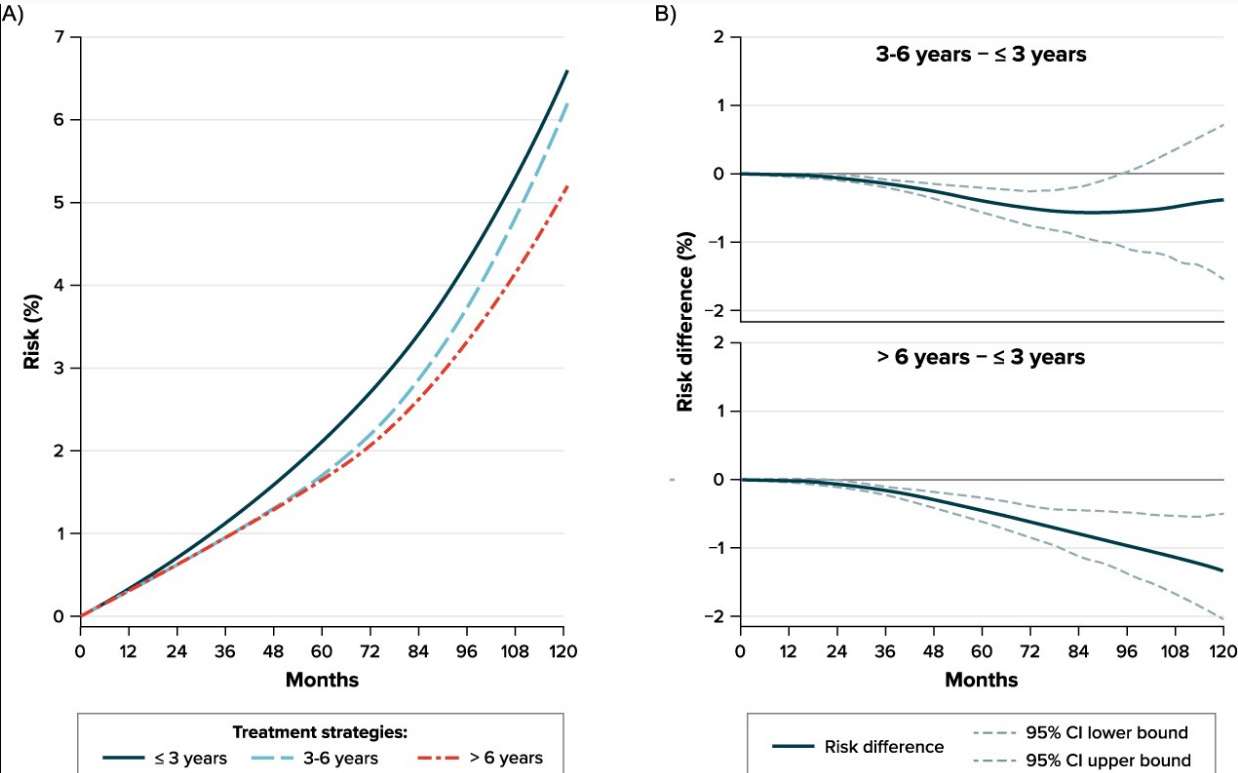
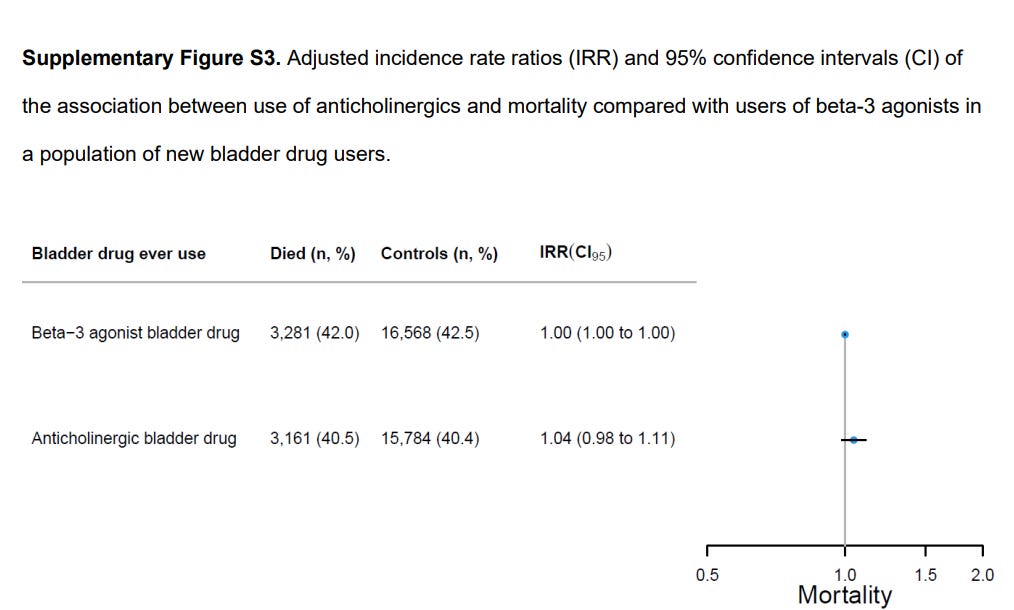
This study is the nail in the coffin on anti-cholinergics and dementia. They compared the effect of anti-cholinergic bladder drugs to a drug that aided in bladder control but had no cholinergic properties — miragebron. In comparison to people who used mirabegron, anti-cholinergic users were less likely to get dementia.
Compared with non-use, ever use of anticholinergic bladder drugs was associated with an increased risk of dementia, with an incidence rate ratio of 1.44 (95% confidence interval (CI) 1.40 to 1.48). The incidence rate ratio increased with increasing cumulative drug use, from 1.31 (95% CI 1.27 to 1.36) for 1-90 defined daily doses to 1.68 (1.59 to 1.76) for >365 defined daily doses. Compared with non-use, all types of anticholinergic bladder drugs were associated with increased incidence rate ratios for dementia: tolterodine 1.43 (95% CI 1.38 to 1.49), solifenacin 1.37 (1.29 to 1.46), trospium 1.52 (1.37 to 1.67), and fesoterodine 1.48 (1.26 to 1.74). The increased risk of dementia with use of anticholinergic bladder drugs was not seen when compared directly with the use of the β3 agonist mirabegron (incidence rate ratio 0.82, 95% CI 0.74 to 0.92), irrespective of the type of anticholinergic drug.
The abstract is a little confusing. Fortunately, their figures are not. They conducted the regular, observational analysis first, and found a positive correlation between anti-cholinergic drug intake and future dementia.
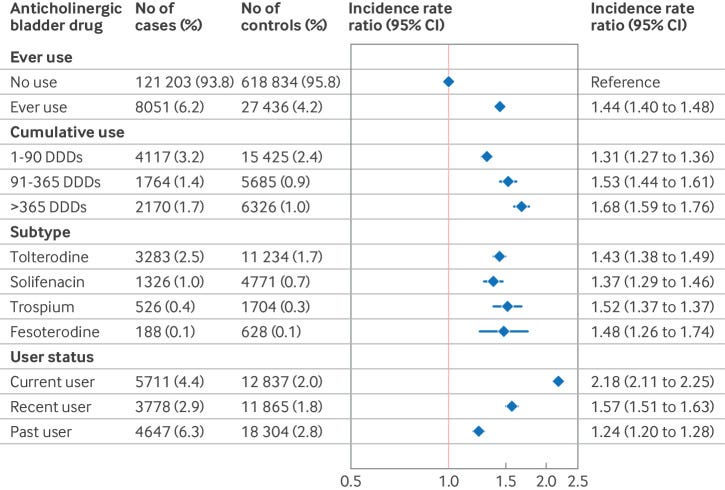
According to this table, using an anti-cholinergic bladder drug for only 1-90 days is associated with a 31% increase in the risk of dementia. If that’s a real effect, surely the power users who took anti-cholinergics daily for 30 years would be totally cooked? What about those teenagers who megadose them for fun?
They then ran the analysis which anti-cholinergic bladder drugs to the non-cholinergic variant, Mirabegron. Anti-cholinergic drugs had a protective effect on dementia.
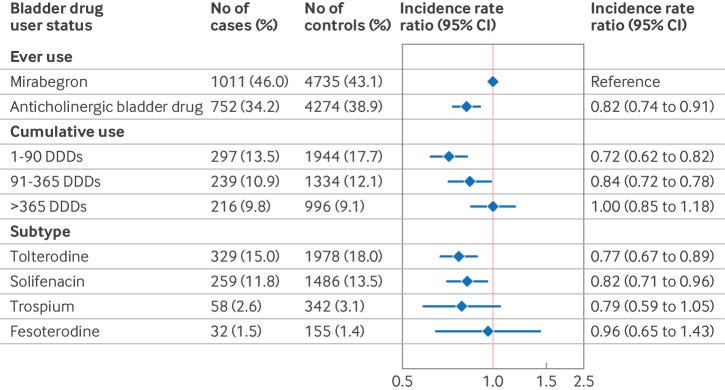
I checked the supplement, and there was no statistically significant difference in all-cause mortality.
Not only are the associations between anti-cholinergic drugs and dementia dependent on the specific drug type, if we evaluate drugs that have been shown to relate to dementia in multiple studies — it turns out it’s still confounding.
Now, we pivot to in-vitro.
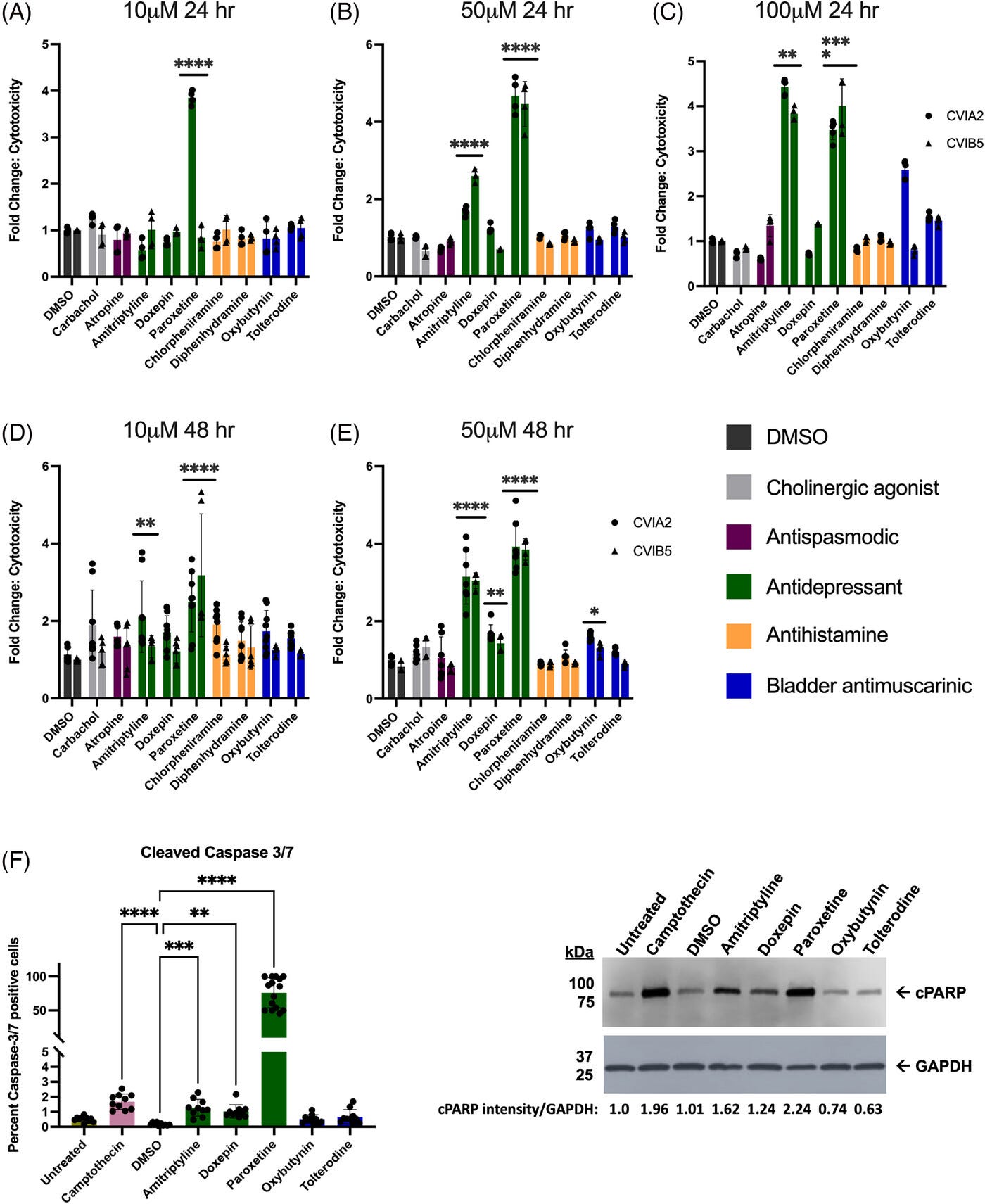
I found one nice study that did it — examining the effects of anti-cholinergic drugs on neurons when they were grown outside of the human body. Theoretically, if they are harmful in humans, we might see their effects take place in artificial cultures as well. It turns out that some of the anti-cholinergic drugs had measurable toxic effects:
We treated human induced pluripotent stem cell (hiPSC)–derived neurons with eight drugs representing different AC medication classes, including antidepressants, bladder antimuscarinics, antihistamines, and antispasmodics. We analyzed these neurons for cytotoxicity, amyloid beta (Aβ) peptide levels in the conditioned medium, and the level of intracellular phosphorylated tau from these cultures.
We observed that antidepressants and bladder antimuscarinics were consistently cytotoxic, whereas antihistamines and antispasmodics did not show overt cytotoxicity at the times and concentrations that we tested. Some of the cytotoxic medications altered the amounts of Aβ1‐42 peptides, but there were no significant differences in the intracellular ratio of phosphorylated tau/total tau between AC drug treatments.
All three anti-depressants showed robust cytotoxic effects (poisonous to cells). One of the anti-muscarinics (bladder control drugs) showed a slight toxic effect, but it might have been a mere statistical fluke:
When it came to the development of amyloidic plaque, which likely contributes to cognitive decline, Oxybutynin (bladder control drug) showed up as a contributor to its formation. They couldn’t detect changes in tau protein formation; in some cases, this might have been because of the toxicity of some of the medications.
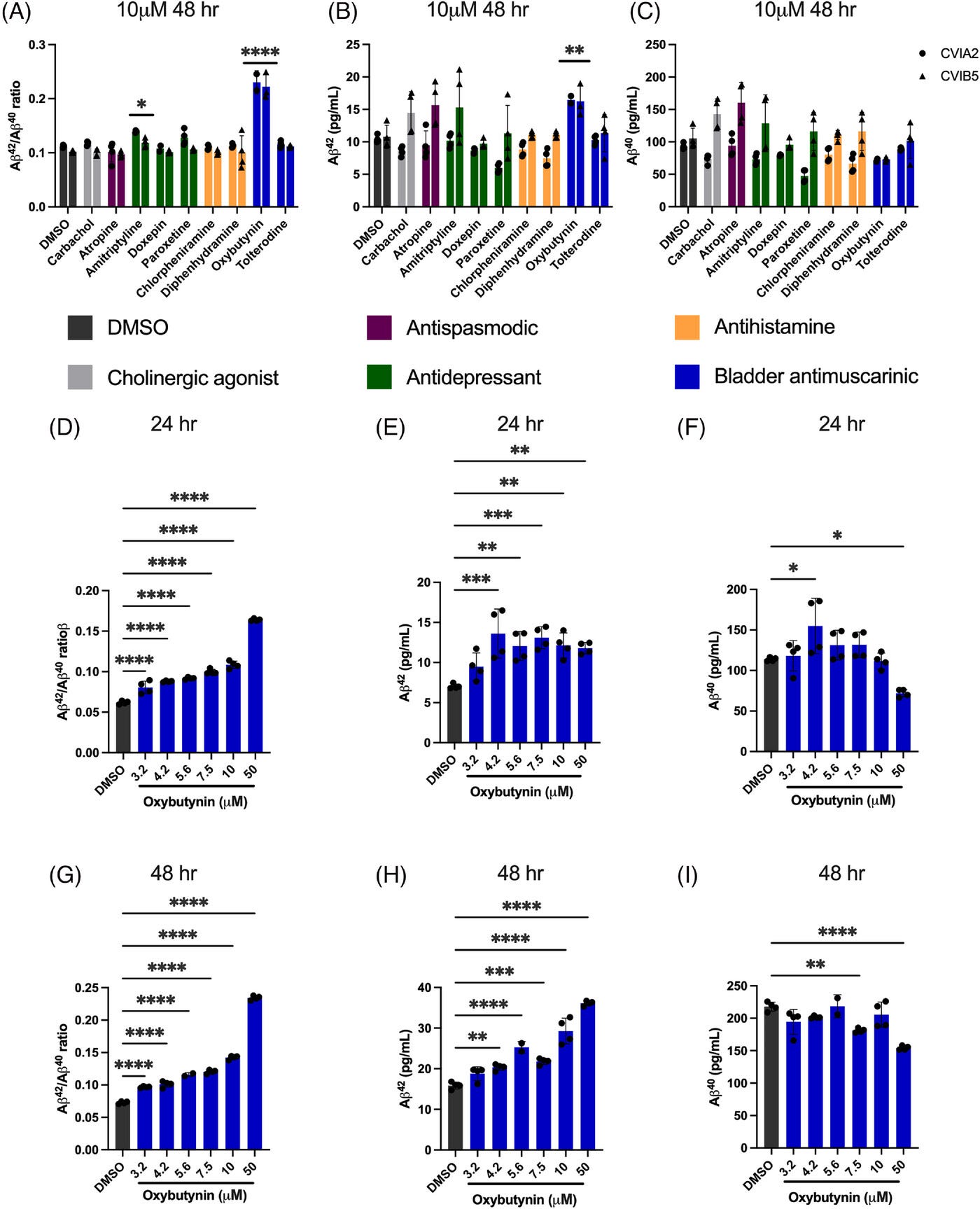
Some of these results in-vitro match the observational study — both methodologies agreed that the antihistamine and antispasmodic drug variants had no measurable effect on cognitive decline. It’s hard to compare the two methods, since they aren’t always using the same drugs, at the same doses.
Besides these studies, I didn’t find much of value: one study that examined effects of anti-cholinergics in 55 year olds on dementia found a p = .04 and a p = .01; an AI prediction study that was able to reliably predict Alzheimer’s diagnoses using medical records, anti-cholinergic drugs only came up in the top 20 factors once (other drugs like benzos came up more often); and some other observational studies that said the same thing.
One could argue that, because cholinergic drugs are meant to treat Alzheimer’s, that means that anti-cholinergics must have the opposite effect. And they are — in the short term. And it’s not like cholinergics are that effective — according to the largest meta-analysis, the effect they have on cognition in people with AD is about 0.33 standard deviations5; similar to the effects of a generic stimulant on cognitive ability.
So
The answer is no, anti-cholinergic drugs do not cause dementia6.
The big problem with the theory is that it is too general. In some cases, anti-cholinergic drugs have protective side effects, like the anti-hypertensives, so it would be crude to hypothesise they are pathogenic. “Don’t eat yoghurt because it has saturated fat”. Some anti-cholinergic drugs don’t even cross the blood brain barrier, so it’s not mechanistically possible for them to cause dementia. I guess it’s great marketing for other drugs that don’t have this effect.
The mechanistic reasoning behind the theory is not irrational, but it’s flimsy in the sense that it relies on believing in the existence and importance of 4th order effects. There’s also reason to think obesity and the cardiovascular system are implicated in dementia, if this is the case, then drugs that affect these systems should naturally affect dementia as well. Also, a general example of why I dislike mechanistic arguments in medicine.
Anti-cholinergics kind of suck as a drug class. They make you tired and dull, but if you take them to sleep, then the quality of your sleep decreases. As such, pharmacists try hard to get these drugs to not cross the blood-brain barrier.
The reason I was interested in this research is that I take benadryl, an anti-cholinergic drug, as a sleep aid. I doubt I’d ever actually develop dementia due to my family history, unless I live to be over 100 years old — that’s when the real decline starts.
Wrote activate for some reason. I have a consistent problem with signs/directions, e.g. in math I would sometimes write -2 instead of 2.
The statistical tests aren’t properly run (you want an interaction here), but the evidence is visually compelling.
Thank mr Cremieux for pointing that out.
One study has a standard deviation of ADAS-Cog of 12.1, 7-10 according to this study, and 5-10 according to this one. I'll guess it’s 8. The abstract of the meta-analysis:
After 26 weeks of treatment, donepezil compared with placebo was associated with better outcomes for cognitive function measured with the Alzheimer’s Disease Assessment Scale‐Cognitive (ADAS‐Cog, range 0 to 70) (mean difference (MD) ‐2.67, 95% confidence interval (CI) ‐3.31 to ‐2.02, 1130 participants, 5 studies), the Mini‐Mental State Examination (MMSE) score (MD 1.05, 95% CI 0.73 to 1.37, 1757 participants, 7 studies) and the Severe Impairment Battery (SIB, range 0 to 100) (MD 5.92, 95% CI 4.53 to 7.31, 1348 participants, 5 studies). Donepezil was also associated with better function measured with the Alzheimer’s Disease Cooperative Study activities of daily living score for severe Alzheimer’s disease (ADCS‐ADL‐sev) (MD 1.03, 95% CI 0.21 to 1.85, 733 participants, 3 studies). A higher proportion of participants treated with donepezil experienced improvement on the clinician‐rated global impression of change scale (odds ratio (OR) 1.92, 95% CI 1.54 to 2.39, 1674 participants, 6 studies). There was no difference between donepezil and placebo for behavioural symptoms measured by the Neuropsychiatric Inventory (NPI) (MD ‐1.62, 95% CI ‐3.43 to 0.19, 1035 participants, 4 studies) or by the Behavioural Pathology in Alzheimer’s Disease (BEHAVE‐AD) scale (MD 0.4, 95% CI ‐1.28 to 2.08, 194 participants, 1 study). There was also no difference between donepezil and placebo for Quality of Life (QoL) (MD ‐2.79, 95% CI ‐8.15 to 2.56, 815 participants, 2 studies).
2.67/8 = 0.33
There are always nuances, but language is about mutual understanding, not essence.


not to be rude, but: was any AI used in the development of this post?
well done piece. I'm still not quite convinced anti-cholinergics do NOT increase dementia risk, but it is not a dramatic effect even if there is one.
I believe that anti-cholinergics dull your thinking and that over time this has a cumulative effect that depresses cognition.
This can lead to dementia symptoms for senior adults whose cognitive load is light, and who are on 10 medications.
I'm not sure the tau tangles theory holds water.
Too many people are perfectly fine in life. When they die, autopsy reveals their brains are full of tau tangles .
I think there's other explanations such as mitochondrial dysfunction that are much better for explaining dementia.